Why vaccination fails against H9N2 avian influenza virus in the field
WIAS Magazine - Summer edition 2022
Research Highlight
H9N2 avian influenza virus (AIV) is the most prevalent and harmful low pathogenicity avian influenza virus (LPAIV) in the world, which not only seriously harms animal husbandry but also poses a huge challenge to public health. Some researchers are concerned that H9N2 may be a spark to the emergence of the next influenza pandemic, either by directly crossing the species barrier or by donating part of its genes to a pandemic virus (1-3). Vaccination is the main strategy for controlling H9N2 AIV in China, Israel, South Korea, Morocco, Pakistan, Egypt, Iran etc. However, most vaccination programs against H9N2 AIV have shown to be ineffective against infection and transmission in the field, although those vaccines were reported to work well in the laboratory. Antigenic distance between vaccine and field strain is considered to be the most important factor to vaccine failure in poultry. However, Sitaras et al (4) proved that when a high immune responses against the challenge strain is present in over 85% of the population of vaccinated chickens, the transmission of AIV (H5) can be stopped, regardless of the antigenic distance. Therefore, in this study, we believed that other factors may play a role and interfere with vaccine efficacy in poultry in the field. We hypothesized that maternal-derived antibodies (MDAs), the transfer of maternal antibodies through the yolk sac, are a possible reasons of vaccination failure in poultry in the field.
To investigate this, we went to a big poultry farm in China and did an epidemiological investigation for three months. We designed an animal experiment in the field.Three groups of chickens were used. Each group had sixteen 1-day-old chickens consisting of half commercial broilers and half Specific Pathogen-Free (SPF) chickens, and one group was housed in one isolator. In Group 1, all chickens were vaccinated subcutaneously in the neck with the inactivated H9N2 vaccine at a dose of 0.1 mL per chicken at day one after hatching. In Group 2, all chickens were immunized with 0.3 mL per chicken of the same vaccine at 21-day-old days of age according to the company’s vaccination procedure in the field. In Group 3, chickens were not vaccinated and used as negative control. Serum samples were collected from each chicken at 0, 7, 14, 21 and 28 days post vaccination (dpv). The hemagglutination inhibition (HI) titers were used to evaluate humoral immunity. The results (Figure 1) showed that in 1-day-old commercial broilers, high HI titers were measured already at day 0 before vaccination, which we attributed to the presence of high titers of MDAs; the titers of vaccinated broilers decreased overtime consistent with the degradation of MDA. In 21-day-old commercial broilers, however, low HI titers were detected at day 0 before vaccination, but HI titers significantly increased after vaccination. When analyzing the humoral responses of SPF chickens (no MDAs), they all mounted high HI titers after vaccination. Hence, these results strongly supported our hypothesis that MDAs may interfere with the humoral immune responses of chickens to vaccination.
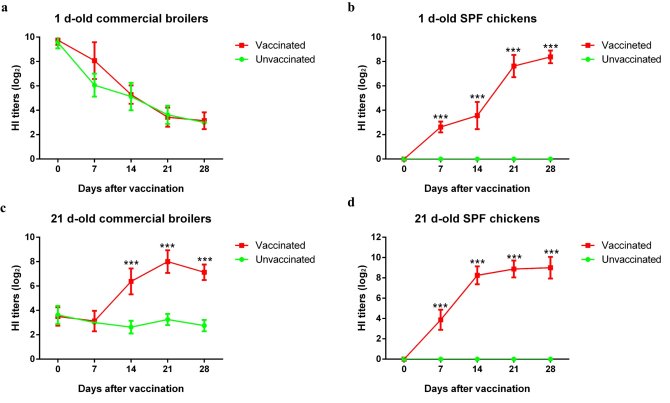
Figure 1. Seroconversion after immunization with the inactivated H9N2 AIV vaccine in commercial broilers and SPF chickens. The symbol (*) denotes differences between two groups at the same time point (* P<0.05, ** P<0.01, and *** P<0.001).
There are some drawbacks of performing experiments with commercial broilers. For instance, broilers may be already infected by some pathogens in the hatchery before the experiments, which may interfere with immune responses as well. Therefore, in order to confirm the results, we mimicked the presence of MDAs in SPF chickens by passively transferred antibodies (PTAs). Four groups of chickens were used, and each group consisted of five 1-day-old SPF chickens. In group 1, chickens without PTAs were vaccinated (positive control). In group 2, chickens with PTAs were not vaccinated (negative control). In group 3, chickens with PTAs from commercial poultry hens were vaccinated, and in group 4 chickens with PTAs from immunized SPF chickens were vaccinated. Chickens in each group were challenged intravenously with 0.1 mL 106 EID50/0.1 mL of homologous H9N2 AIV at 28 dpv. Oropharyngeal and cloacal swabs were collected at 3- and 5-days post-challenge, respectively. The results confirmed that chickens with high PTAs titers could not develop high HI titers after vaccination (Figure 2a). Furthermore, vaccinated chickens with high PTAs titers still shed virus after challenge with homologous H9N2 virus, but vaccinated chickens without PTAs didn’t shed virus at all after challenge (Figure 2b). Interestingly, after challenge, vaccinated chickens with high PTAs titers mounted faster and higher immune responses compared with unvaccinated chickens (Fig 2c).
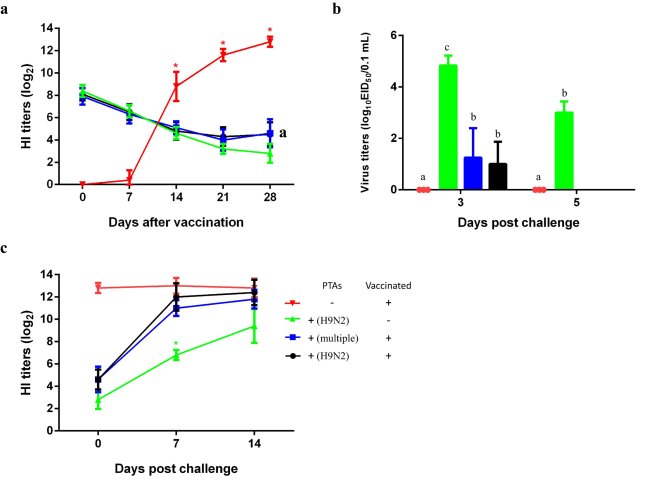
Figure 2. Mimicking MDAs with PTAs in SPF chickens and the responses to
vaccination and challenge. Seroconversion after vaccination (a), viral titers
in oropharyngeal swabs after challenge (b) and seroconversion after challenge
(c) are shown in Fig. 2. Superscripts a-c denote differences among each group
at the same time point. The symbol (*) denotes differences among groups at the
same time point. P < 0.05 was considered significant.
To identify the effect of different PTAs titers, we designed another animal experiment. Four different titers of antibodies were transferred into 1-day-old SPF chickens (n = 10), and then half of these chickens from each group were vaccinated with 0.1 mL of inactivated H9N2 vaccine per chicken one day later (Group 1: HI = 12 log2; Group 2: HI = 9 log2; Group 3: HI = 6 log2; Group 4: HI = 0 (PBS)). Then, all chickens were vaccinated with 0.2 mL of vaccine fourteen days later as a booster. Serum samples were collected from each chicken at 0, 7, 14, 21 and 28 dpv. The results (Figure 3) showed that high (HI = 12 log2) and medium (HI = 9 log2) PTAs titers indeed interfered with the development of antibodies after the first vaccination (at least up to 21 days post vaccination), while low (HI = 6 log2) titers of PTAs and PBS did not. In the groups that received high and medium PTAs titers and a booster vaccination, the antibody titers developed faster than in chickens that received high and medium PTAs titers and no booster vaccination.
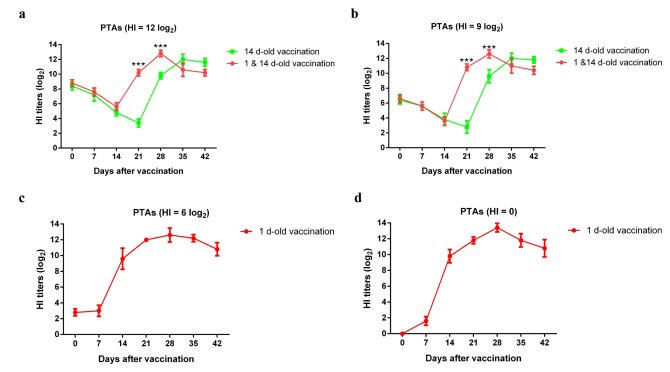
Figure 3. Seroconversion to prime or booster vaccination in SPF chickens with high, medium or low titers of PTAs. The symbol (*) denotes differences between two groups at the same time point (* P<0.05, ** P<0.01, and *** P<0.001).
Overall, the present study suggests that H9N2-specific MDAs are one of the factors contributing to inactivated H9N2 vaccine failure in poultry in the field. High and medium titers of MDAs interfered with the humoral immune responses in broilers, but a booster dose induced faster and higher humoral immune responses, even under the condition of MDA interference. Our results highlighted that we should pay more attention to MDAs interference on influenza virus vaccines in the field instead of only antigenic distance. It is important to create news vaccines to overcome MDAs interference in the field. The current manuscript is based on the articles published in “Animal Diseases”, DOI: 10.1186/s44149-022-00040-0.
References
1. Caceres CJ, Rajao DS, Perez DR. Airborne Transmission of Avian Origin H9N2 Influenza A Viruses in Mammals. Viruses. 2021;13(10).
2. Peacock THP, James J, Sealy JE, Iqbal M. A Global Perspective on H9N2 Avian Influenza Virus. Viruses. 2019;11(7).
3. Rahimirad S, Alizadeh A, Alizadeh E, Hosseini SM. The avian influenza H9N2 at avian-human interface: A possible risk for the future pandemics. Journal of Research in Medical Sciences the Official Journal of Isfahan University of Medical Sciences. 2016;21(1):51.
4. Sitaras I, Rousou X, Kalthoff D, Beer M, Jong MCMD. Role of vaccination-induced immunity and antigenic distance in the transmission dynamics of highly pathogenic avian influenza H5N1. Journal of the Royal Society Interface. 2016;13(114):20150976.