Project
LymphChip: Developing immunocompetent human organ-on-a-chip models with integrated lymph drainage
Organ-on-chip (OoC) models revolutionize drug discovery, yet current versions lack the lymphatic and immune systems crucial for understanding health and disease. This NWA proposal aims to enhance OoC models by incorporating immunocompetent human cell cultures, advancing our understanding of disease, drugs, and tissue repair mechanisms. This innovative approach seeks to replace animal models and provide new models for (personalized) medicine.
Background
The lymphatic and immune systems are big players in our health, impacting diseases and how well drugs work. They help maintain fluid balance, keep an eye on the immune system, and aid in healing tissues. But, current lab models don't consider these systems much, making it hard to develop new drugs. The solution would be to use (stem cell-derived) organ-on-chip setups, mimicking real organs like the heart (focus at VUM), gut (focus at WUR), and skin (focus at LUMC), in which we add lymph and immune cells. This approach allows an in vitro combination of the missing pieces making the models more like our bodies. The goal is to make drug discovery more accurate and personalized using these models.
The lymphatic and immune systems are big players in our health, impacting diseases and how well drugs work. They help maintain fluid balance, keep an eye on the immune system, and aid in healing tissues. But, current lab models don't consider these systems much, making it hard to develop new drugs. The solution would be to use (stem cell-derived) organ-on-chip setups, mimicking real organs like the heart (focus at VUM), gut (focus at WUR), and skin (focus at LUMC), in which we add lymph and immune cells. This approach allows an in vitro combination of the missing pieces making the models more like our bodies. The goal is to make drug discovery more accurate and personalized using these models.
Our immune system is like a superhero defending us, but sometimes it messes up, causing diseases. Imagine models that capture this superhero's moves! Current models miss these actions, making them less helpful in developing medicines. Our plan is to create super realistic setups where these superhero actions are considered. By adding lymphatic and immune bits into the mix, we're hoping to revolutionize how we understand diseases and fix our bodies. It's like upgrading from black and white to technicolor in the world of medicine!
Dysfunction in the immune and lymphatic systems poses significant healthcare challenges, often overlooked in preclinical models. Lymphatic dysfunction is implicated in numerous diseases, influencing the efficacy of drugs and stem cell therapies. Vital for fluid balance and immune processes, the lymphatic system regulates immune cell movement, surveillance, and responses. It is increasingly recognized for its role in tissue regeneration. Comprising lymph vessels and filtering nodes, it returns fluid and immune cells to circulation. Lymph nodes initiate adaptive immune responses crucial for lifelong protection.
Current in vitro models lack lymphatics, hindering the recognition of side effects from therapies affecting these systems. Urgent calls from society, academia, and industry highlight the need for research models addressing lymphatic and immune roles, as mice, while informative, differ significantly from humans in immune cell behavior. Ethical concerns and the 3Rs (reduction, refinement, replacement) emphasize the necessity for human-relevant organotypic models to replace animal testing in drug development. Incorporating human immune components into disease and regenerative medicine models is crucial for advancing personalized and precision medicine.
Advancements in engineering microphysiological systems, particularly through stem cell-derived organoids, have enhanced physiological realism, yet none fully integrate immune cells and lymphatic vasculature. Existing models lack 3D structures for lymph fluid and immune cell interaction. Organ-on-Chip (OoC) technology is promising, incorporating tissue-specific cells and biophysical features like fluidic flow and strain. OoC models, populated by organoids or mini-organs from human induced pluripotent stem cells (hiPSC), present advantages for isogenic stromal cells. However, the critical inclusion of lymphatic and immune systems in OoC models is vital to accurately capture human disease and tissue repair mechanisms, marking an essential frontier in research.
Main goal and choice of model systems
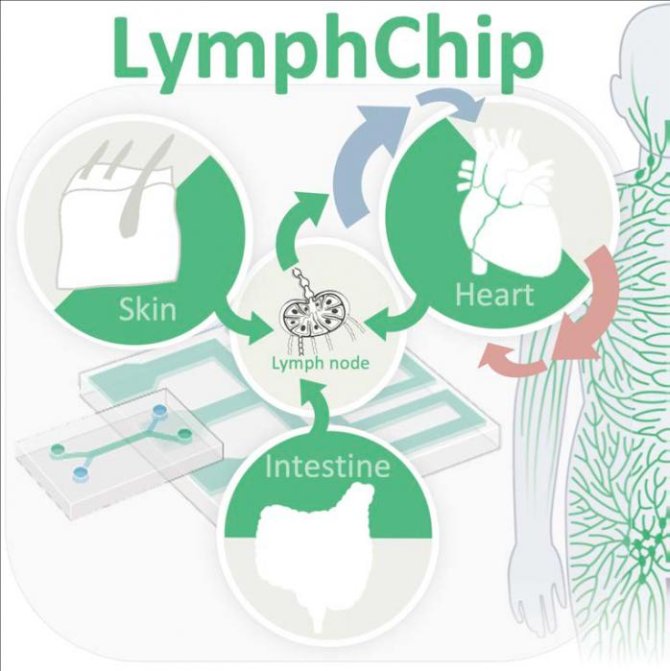
Our goal is to create three tissue models (heart, intestine and skin) containing a 3D lymphatic system able to undergo fluidic flow in an OoC format with companying immune cells derived from human stem cells. The result will be healthy and diseased human tissue models with unprecedented realism through incorporation of functional lymphatics. We have chosen these three tissues to cover the diverse roles of lymphatics in tissues with different developmental origins (see Table below and Fig. 1). Without lymphatics and immune cells that are tissue specific, many in vitro models will fail to capture tissue reality in human disease. Technologies developed for these three tissues can be easily adapted to other healthy and disease organ systems in the future (e.g. liver, kidney), paving the way for a generation of superior immunocompetent human cell culture models with integrated lymph drainage and immune cells. On the other hand, there are many similarities and shared challenges through which the three models will reinforce each other synergistically. For example, lymphatics drain (excessive) interstitial fluid and immune cells from all tissues under different (patho)physiological conditions thus preventing oedema and inflammation.
Objectives
We will achieve our aim through four objectives:
- develop heart, intestine and skin OoCs with integrated lymphatics and immune cells;
- test whether the immune competent organ-lymph models are functional;
- develop quantitative readouts of healthy and disease states including real-time (dynamic) imaging and biomarkers;
- test a panel of clinically relevant drugs to qualify tissue OoCs with lymphatics and immune cells in preparation for regulatory acceptance as a validated animal alternative.