Trypanosoma carassii, a model for whole host-pathogen study
WIAS Magazine - Summer edition 2023
Research Light
What is a parasite?
An organism that feeds strictly at the expense of a host organism of a different species, either permanently or during a phase of its life cycle. Within the vast diversity of parasites, we are particularly interested in one specific type, Trypanosomes, in Wageningen.
Trypanosomes are motile flagellated unicellular organisms capable of infecting a wide range of living organisms, including plants and animals.
Amongst the hosts of Trypanosomes are humans, who are infected by two subspecies, Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense. These parasites are still present in Africa where, they are actively transmitted by a vector, the tsetse fly, and cause a disease named Human African Trypanosomiasis (HAT), formally called sleeping sickness (Figure 1). Despite a WHO statement declaring that trypanosome transmission will be interrupted by 2030, scientists don’t have much confidence that such progress can be achieved. Indeed, several eradication attempts have already been made without success. The socio-political context, which includes political insecurity and environmental degradation, makes these predictions unlikely. This parasite can evolve rapidly and has already developed resistances to numerous currently used drugs. It is therefore essential to continue research efforts on this specific host-pathogen interaction with the ultimate aim to fight infections through large scale drug treatment programmes and possibly vaccination leading to eradication.
What is sleeping sickness?
HAT is characterized by the presence of trypanosomes in the blood and tissues following the bite of an infected tsetse fly. After a period of time, the parasite is found in the central nervous system. The early symptoms of this disease can be mild but the final phase of the disease, known as the encephalic stage is caused by the presence of the trypanosomes in the brain. At this stage, the patient develops sleep disorders with a reversal of the circadian cycle, behavioural disturbances, and motor problems, and death due to cachexia.
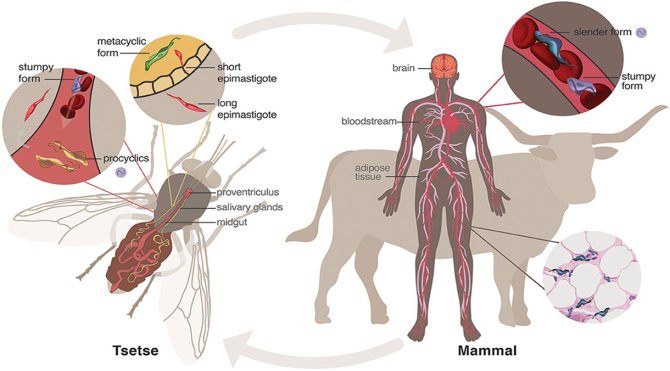
Figure 1. African trypanosomiasis cycle with the tsetse vector and different host. The Tsetse fly bites the host and transmits metacyclic preadapted parasites that divide in the blood of the host as slender trypomastigote. The cycle continues when this contaminated fly bites another host and transmit the parasite (1).
Which parasite is the most studied?
Trypanosoma brucei is a highly studied parasite because its subspecies can infect both humans and animals. The model for human studies is Trypanosoma brucei brucei, a parasite found in African cattle herds.
Which questions remain unanswered?
How the trypanosomes enter the brain and the mechanisms for generalized tissue invasion in humans remain largely unknown. One of the main obstacles is the inability to visualize the trypanosome's passage into the brain. No hypothesis has convinced the scientific community so far. To visualize and understand how the parasite enters the brain, a transparent living organism model would be a major asset. For this particular question, the resources and models established by Dr. Maria Forlenza's team in the AFI group at WUR are invaluable (2), (3).
What is the plan?
In the AFI team, the trypanosome studied is Trypanosoma carassii, a species capable of infecting freshwater fish, such as common carp (Cyprinus carpio), goldfish (Carassius auratus), and other cyprinid fish. With the help of the Department of Biochemistry at Cambridge University, we have established transgenic lines of T. carassii, expressing fluorescent proteins. These genetically modified parasites can be more easily observed in transparent fish, such as albino zebrafish or transparent zebrafish larvae. The objective of my work here is to explore parasite invasion in fish in order to understand the mechanisms employed by the parasite to invade and multiply in tissues - a true observational approach aimed to capture crucial moments in the course of the infection (Figure 2). Fluorescent parasites are more easily traceable with a microscope, and we seek to track them to describe their behaviour as accurately as possible. The results of this research could provide insights into the pathophysiology of the human disease and help understand the mechanisms of brain invasion in humans.

Figure 2: Representation of process of infection and imagery.
First results
We made different lines of parasites expressing different fluorescents proteins such as mNeonGreen (mNG) or Ruby3 proteins (Figure 3a). We performed transgenesis successfully and we currently infect larvae with a known number of parasites to track them while the fish is alive, describing the course of the infection (Figure 3b).

Figure 3: The two steps process of Trypanosoma carassii visualisation in
zebrafish larvae. a: In vitro studies: Transgenesis on Trypanosoma carassii. b: In vivo studies: Parasite distribution in the trunk of transgenic zebrafish larvae. Zebrafish larvae marking the endothelium in red (Tg(Flk:mCherry)Flk=Vegfr2=kdrl) were infected with mNG Trypanosoma carassii (green) and imaged at the indicated time points after fixation with 1% PFA.
Conclusion: We successfully generated transgenic Trypanosoma carassii expressing fluorescent proteins that are easy to track in vivo in live zebrafish larvae. We have great expectations that this tool can help us better understand the interaction between the host immune system and the parasite. At the end some of these observations could be interesting to extrapolate to cattle and humans to finally understand how trypanosomes enter the brain of their host and cause death.
References :
1. Rijo-Ferreira F, Takahashi JS. Sleeping Sickness: A Tale of Two Clocks. Front Cell Infect Microbiol [Internet]. 2020 [cité 8 juin 2023];10. Disponible sur: https://www.frontiersin.org/articles/10.3389/fcimb.2020.525097
2. Dóró É, Jacobs SH, Hammond FR, Schipper H, Pieters RP, Carrington M, et al. Visualizing trypanosomes in a vertebrate host reveals novel swimming behaviours, adaptations and attachment mechanisms. Clayton C, Akhmanova A, éditeurs. eLife. 24 sept 2019;8:e48388.
3. Jacobs SH, Dóró E, Hammond FR, Nguyen-Chi ME, Lutfalla G, Wiegertjes GF, et al. Occurrence of foamy macrophages during the innate response of zebrafish to trypanosome infections. McConville MJ, Rothlin CV, McConville MJ, De Trez C, éditeurs. eLife. 11 juin 2021;10:e64520.