
Project
Long-term phosphate fertilization trials
Acquiring optimal crop yields is not possible without phosphorus (P) fertilisation because P is an essential element of biomolecules in plants. Since P reacts strongly with various soil components, like iron- and aluminum-oxides, calcium-carbonates, and clay mineral edges, the effectivity of P fertilisation in relation to crop yields and P uptake depends strongly on soil composition. Here, I will study how the fate of P from applied mineral P fertiliser and how crop yields and P uptake differ between calcareous loam soil and acid sandy soil. This knowledge will help to improve a judicious usage of mineral P fertiliser in the future.
Background
Improving the P status of soil by fertilisation is a delicate balance to avoid P limitation for crop uptake on the one hand while limiting P losses to the environment on the other hand. For example, 43% of agricultural land in the Netherlands is estimated to exceed the environmentally acceptable soil P status threshold due to excessive P surplus applications via animal manure in the past decades. Moreover application of P beyond crop demands is a waste of a critical resource, although phosphate reserves are still predicted to last for >300 years. Current P fertilisation recommendations rarely consider differences between soil types regarding P retention by reactive soil components, while P binding processes in soil vary depending on pH and soil contents of iron- and aluminium oxides, clay, calcium carbonate, and soil organic matter. A better understanding of the effects of relevant soil properties on the relation between P fertilisation rate ~ soil P status ~ crop yield will helps to come to a more efficient usage of mineral P fertiliser.
Description
In this project, I study how P sorption/precipitation processes and the relation between SPTs1 and crop yield differs between calcareous loam soil and acid sandy soil.
I do this by using currently available data and new chemical analyses on (archived) soil samples from two arable long-term P fertilisation trials which started in 1971 and which are still in use. One experiment is situated near the village of Marknesse on a calcareous loamy soil and one experiment is near Wijster on an acid sandy soil. Both experiments have been fertilised with superphosphate. Natural rock phosphate is used as an additional P source in the experiment near Wijster.
Regarding soil P chemistry, an important point is to elucidate to what degree calcium-phosphate precipitation is of quantitative importance in comparison to P sorption to iron and aluminium oxides and to what degree the applied mineral P fertiliser is lost to stable P fractions, e.g., mineral phosphate precipitates or P strongly fixed in the interior of oxides.
Regarding the connection between SPTs and crop yield, a major point of focus is whether SPTs give a comparable indication of soil P availability in the two contrasting soils types or otherwise how to correct for differences in critical SPTS between soils (Figure 1).

Different measurement techniques will be used to understand soil P speciation of fertilised soils, ranging from wet chemical extractions to solid P speciation measurements with XANES2.
1: Soil P tests. Wet chemical extractions of soil extracting an operationally defined P pool.
2: X-ray absorption near edge structure. A spectroscopic technique to measure to which atom P is bound.
Some preliminary results
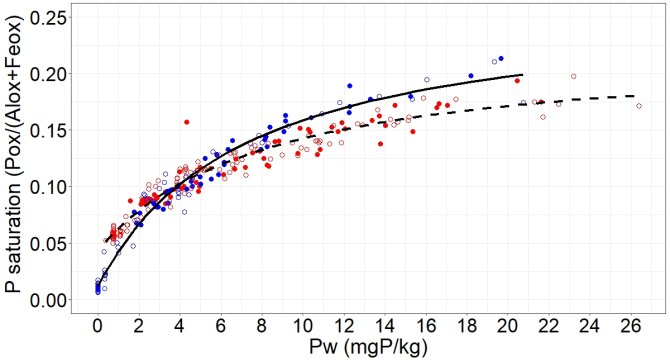
Firstly, soil P saturation (calculated here as acid ammonium oxalate-extractable P divided by the sum of ammonium oxalate-extractable Fe and Al) gives a quite similar prediction of P in soil solution for both the calcareous soil and the acid soil. The oxide content therefore seems to control dissolved P in solution at both locations (Figure 2).
Secondly, after 50 years of P fertilisation of the calcareous soil, 35% of applied P can only be quantified using a method able to determine total soil P, not acid ammonium oxalate as a proxy for the total amount of reversible sorbed P. Hence, 35% of the applied P may have precipitated in the form of calcium phosphates on the calcareous soil, but XANES measurements are still needed to get better insight in the formation of calcium-phosphates.
Lastly, critical SPT values to prevent >10% yield loss are 3x higher at the acid sandy soil than at the calcareous loam soil. Hence, a higher apparent chemical soil P availability is needed for the sandy soil than for the calcareous soil to ensure sufficient P uptake. Pot experiments are planned to be performed to study whether the soil characteristics of the calcareous soil allow for a higher P diffusion rate from the bulk soil solution to the root surface where P uptake occurs, which might explain the lower critical SPTs for this specific soil.