Project
3D electroformed Ni-based electrodes for hydrogen generation and novel electrochemical processes: Characterization and Design
Hydrogen is considered the most promising fuel for its high energy density, abundance, and no emissions during combustion. Today, however, it is almost
entirely produced via steam methane reforming, a process which uses natural gas to produce syngas – H2 mixed with CO2. To avoid the use of natural gas, a non-renewable hydrocarbon, and the production of a greenhouse gas CO2, hydrogen can instead be sustainably produced by water electrolysis using renewable electricity.
Technological challenge
The performance of electrolysis depends heavily on the electrodes, which need to be highly catalytic to facilitate the gas’ formation. Platinum group metals have outstanding catalytic performance but are expensive. Therefore, new cost-effective alternatives are necessary. Since the development of electrolysers, nickel-based materials have remained state of the art non-noble hydrogen evolution reaction (HER) catalysts for alkaline water electrolysis. While nickel is the most active non-noble metal, it does not outperform platinum group metals. Thus, much effort must be put into optimizing nickel-based catalysts’ chemical structure and morphology.

Research goals
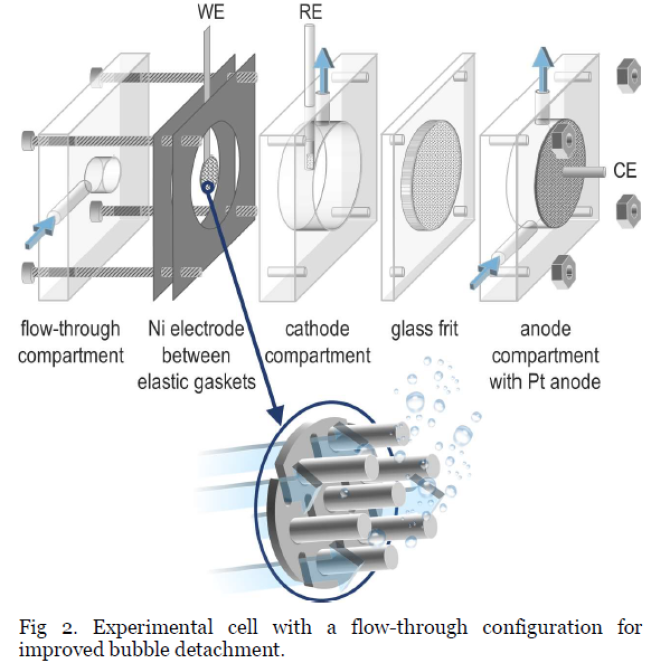
This project investigates pillared nickel electrodes and optimizes their design for H2 production via alkaline water electrolysis.
Focus is put on three research objectives:
1. Testing the electrodes against state of the art alkaline HER catalyst (Raney nickel) and optimizing the electrode design (pillar spacing and length, electrode porosity) for improved performance.
2. Improving the electrodes’ catalytic activity with noble and non-noble dopants.
3. Identifying and demonstrating novel applications for such electrodes.