
Equipment
STREAK camera
Cuvet based time-resolved fluorescence spectroscopy with the streak camera, where changes in fluorescence spectra can be monitored with a time resolution of a few picoseconds. It can measure the dynamics of ultrafast events in photosynthesis, the solvation dynamics of proteins and FRET using the ultrafast rise and decay correlation.
Cuvet based ultrafast time-resolved fluorescence spectroscopy can be performed with the streak camera, where changes in fluorescence spectra can be monitored with a time resolution of a few picoseconds. It can measure the dynamics of ultrafast events in photosynthesis, the solvation dynamics of proteins and FRET using the ultrafast rise and decay correlation.
Background:
After absorption of light, a molecule is in one of its excited states. It can now undergo vibrational relaxation, internal conversion, solvent relaxation, energy transfer and/or fluorescence.

All processes have their own rate (in s-1), and consequently all intermediate states have the own lifetime. When such an intermediate state can fluoresce, the rate of the population of the state (ingrowth) and depopulation (decay) can measured. Each state can be distinguished by its emission spectrum.
The Streak camera set-up can measure these changes with picosecond time resolution and sub-nm wavelength resolution. First, the wavelengths of the fluorescence are spatially separated (horizontal axis) by a spectrograph. This spectrum is projected on the linear input cathode of a streak camera. In the streak tube, the temporal profile is transformed into a spatial profile (vertical axis) combination.
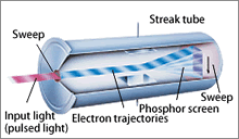
In our set-up, high energy femtosecond pulses from a Ti:sapphire laser pumped regenerative amplifier (RegA) system are wavelength converted by an optical parametric amplifier (OPA/SHG). This results in 250 fs pulses of 400 nm (SHG) or tuneable between 500-700 nm (OPA) with an average power of 20 mW and a 250 kHz repetition rate. Various sample holders are available.

Key specifications:
Available excitation wavelengths: 400, 500-700 nm
Repetition rate: 250 kHz
"Spectrograph gratings:
- 50 gr/mm, blazed 600 nm, width 260 nm,
- 40 gr/mm, blazed at 400 nm, width 320 nm
- 40 gr/mm, blazed at 500 nm, width 320 nm
Wavelength resolution: <1 nm
Time-resolution: 5 ps FWHM instrument response
Publications
- Chukhutsina, V.U., Buchel, C. & Amerongen, H. van (2013). Variations in the first steps of photosynthesis for the diatom Cyclotella meneghiniana grown under different light conditions. Biochimica et Biophysica Acta. B, Bioenergetics, 1827(1), 10-18.
- Laptenok, S., Stokkum, I.H.M. van, Borst, J.W., Oort, B.F. van, Visser, A.J.W.G. & Amerongen, H. van (2012). Disentangling picosecond events that complicate the quantative use of the calcium sensor YC3.60. The Journal of Physical Chemistry Part B: Condensed Matter, Materials, Surfaces, Interfaces & Biophysical, 116(9), 3013-3020.
- Tian, L., Gwizdala, M., Stokkum, I.H.M. van, Koehorst, R.B.M., Kirilovsky, D. & Amerongen, H. van (2012). Picosecond Kinetics of Light Harvesting and Photoprotective Quenching in Wild-Type and Mutant Phycobilisomes Isolated from the Cyanobacterium Synechocystis PCC 6803. Biophysical Journal, 102(7), 1692-1700.
- Tian, Lijin, Stokkum, I.H.M. van, Koehorst, R.B.M., Jongerius, A., Kirilovsky, D. & Amerongen, H. van (2011). Site, rate and mechanism of photoprotective quenching in cyanobacteria. Journal of the American Chemical Society, 133(45), 18304-18311.
- Wientjes, E., Stokkum, I.H.M. van, Amerongen, H. van & Croce, R. (2011). Excitation-energy transfer dynamics of higher plant photosystem I light-harvesting complexes. Biophysical Journal, 100(5), 1372-1380.
- Wientjes, E., Stokkum, I.H.M. van, Amerongen, H. van & Croce, R. (2011). The role of the individual Lhcas in photosystem excitation energy trapping. Biophysical Journal, 101(3), 745-754.
- Koehorst, R.B.M., Laptenok, S., Oort, B.F. van, Hoek, A. van, Spruijt, R.B., Stokkum, I.H.M. van, Amerongen, H. van & Hemminga, M.A. (2010). Profiling of dynamics in protein-lipid-water systems: a time-resolved fluorescence study of a model membrane protein with the label BADAN at specific membrane depths. European Biophysics Journal, 39(4), 647-656.
- Oort, B.F. van, Eremeeva, E., Koehorst, R.B.M., Laptenok, S., Amerongen, H. van, Berkel, W.J.H. van, Malikova, N.P., Markova, S.V., Vysotski, E.S., Visser, A.J.W.G. & Lee, J. (2009). Picosecond Fluorescence Relaxation Spectroscopy of the Calcium-Discharged Photoproteins Aequorin and Obelin. Biochemistry, 48(44), 10486-10491.
- Oort, B.F. van, Murali, S., Wientjes, E., Koehorst, R.B.M., Spruijt, R.B., Hoek, A. van, Croce, R. & Amerongen, H. van (2009). Ultrafast resonance energy transfer from a site-specifically attached fluorescent chromophore reveals the folding of the N-terminal domain of CP29. Chemical Physics, 357(1-3), 113-119.