Longread
Watch/Let Paint dry in different functional layers
Watching paint dry, a proverb that we use if something is boring and takes a long time. However the processes during the drying of paint are essential in many applications. Many surfaces need to be painted or coated to protect it from the environment and to provide a color or a gloss/shine. To provide these different qualities most often different layers of paint/coatings are needed. Take for example the coating on a car as shown in figure 1 at the right. Many layers are needed with their own property to properly to provide the total layer with the correct functionalities.
At this moment each layer of paint need to be added separately. You can image that this takes much time and energy. Therefore, we like to develop a paint that can separate into multiple layers during evaporation.
We can do this by mixing the different paint beforehand. Paints are colloidal suspensions of different particles. The most prominent particles are latex particles (sort of plastic spheres). They provide attachment to the substrate once the paint is dried. By adding another type of particle for example silica particles (kind of very small glass spheres), we could create a paint consisting out of a more soft attachment layer, due to the latex particles and the more hard layer due to the silica particles.
If we make sure the size between these particles is different we can use a specific process called stratification. Stratification occurs during the evaporation of the paint. Both, small silica particles and the large latex particles will accumulate at the evaporation interface, since they are too big to diffuse away from the descending evaporation interface. This gives a concentration gradient in the evaporating paint mixture. The smaller silica particles like to move away from this high concentration, this creates a diffusiophoretic force. This will draw also the large particles along. Since their size is larger than that of small particles they will experience this force more than the small particles. Therefore the large particles are pushed away from the evaporation front, while the small particles will more and more accumulate over there. In this way a paint layer can be formed with more silica particles at the top and more latex particles near to the substrate. (see figure 2)
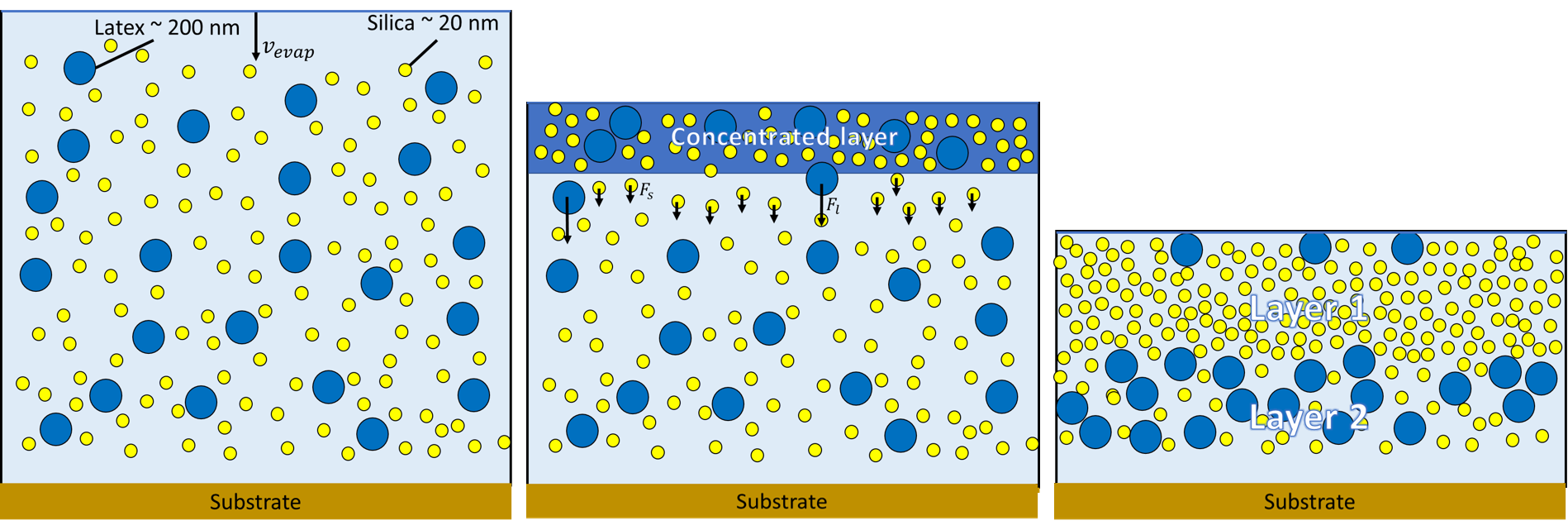
In this research we like to measure and tune this stratification, with different particles and see the effect of for example different particles sizes, evaporation speed, temperature, humidity etc.
- Use Raman confocal spectroscopy to measure stratification and see how the different parameters influence the stratification. (see Figure 3)

Use fluorescently labeled particles to measure the stratification during evaporation with confocal/multiphoton spectroscopy.

- Use simulations on mixtures of spherical particles and rod like particles. Do we still get stratification? Do we see other interesting effects? (see Figure 5)

Please contact me if you are interested via ellard.hooiveld@wur.nl